 What are Atomic Orbitals?Ītomic orbitals are the three-dimensional space near the nucleus of an atom where the possibility of discovering an electron is maximum. The orbitals of an atom are organized into layers or electron shells. The fifth d orbital is fashioned like a long dumbbell with a doughnut in the center. 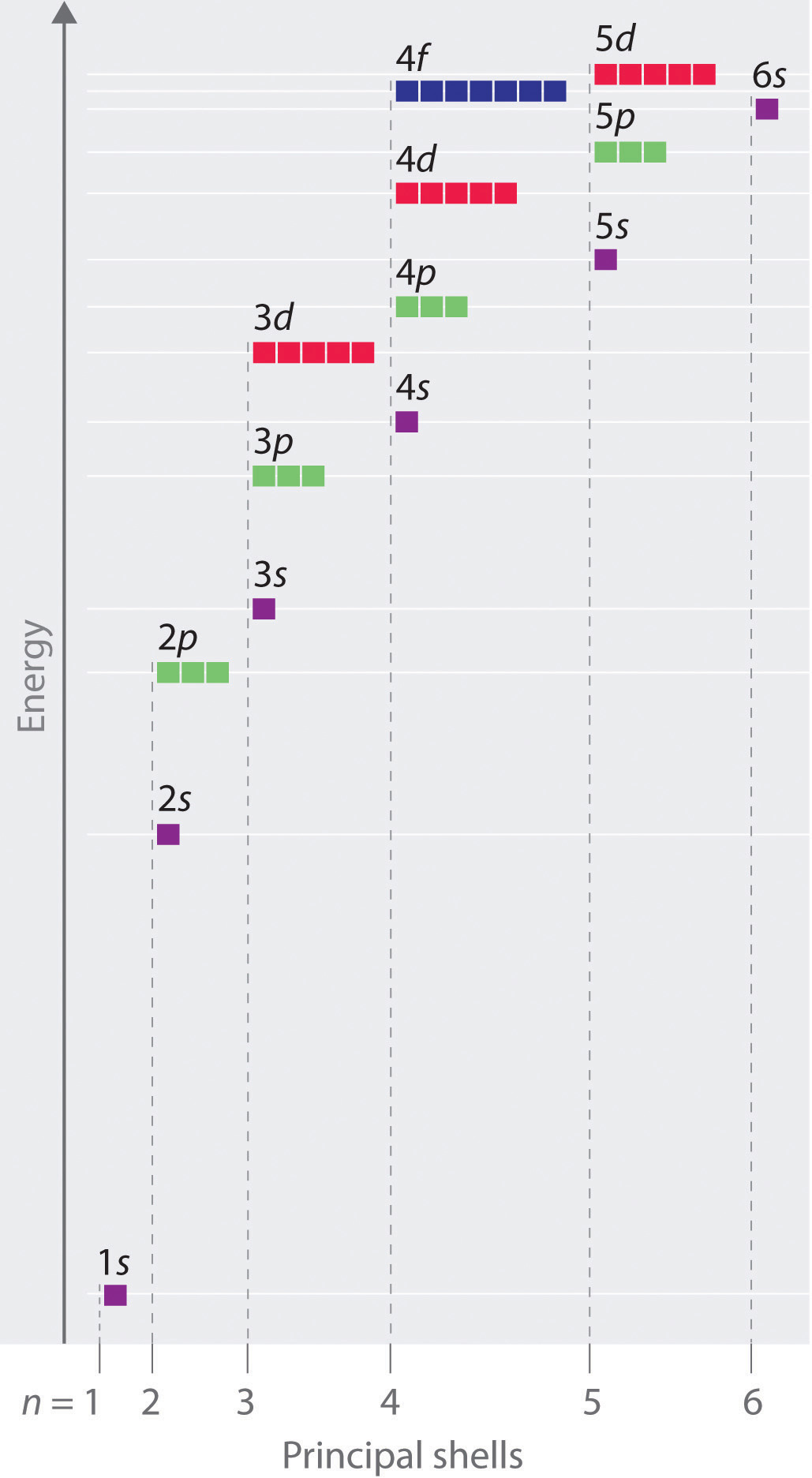
An s-orbital has a spherical nucleus in the center, a p-orbital is dumbbell-shaped, and four of the five d orbitals are cloverleaf shaped. The s and p orbitals are taken into account because they are the most abundant in chemical and biological chemistry. There are four types of orbitals, each with a different shape and represented by the letters s, p, d, and f. The different shapes of atomic orbitals describe that there is either the highest or zero probability of finding an electron around the nucleus in some directions. Atomic orbitals have different shapes according to their type. Augmented Assignment Operators in PythonĪtomic orbitals, which are mathematical functions, describe the wave behavior of electrons (or electron pairs) in an atom.Class 11 NCERT Solutions - Chapter 7 Permutations And Combinations - Exercise 7.1.Importance of Chemistry in Everyday Life.Therefore, for n 1, there is but one atomic orbital and one electron density distribution. Difference Between Mean, Median, and Mode with Examples For every value of the energy E n, for the hydrogen atom, there is a degeneracy equal to n 2.What is the Difference between Interactive and Script Mode in Python Programming?.Molecular Nature of Matter – Definition, States, Types, Examples Hydrogen orbitals or atomic orbits have the most basic or elementary configuration of the electrons placed into the orbitals around the nucleus in an atom. 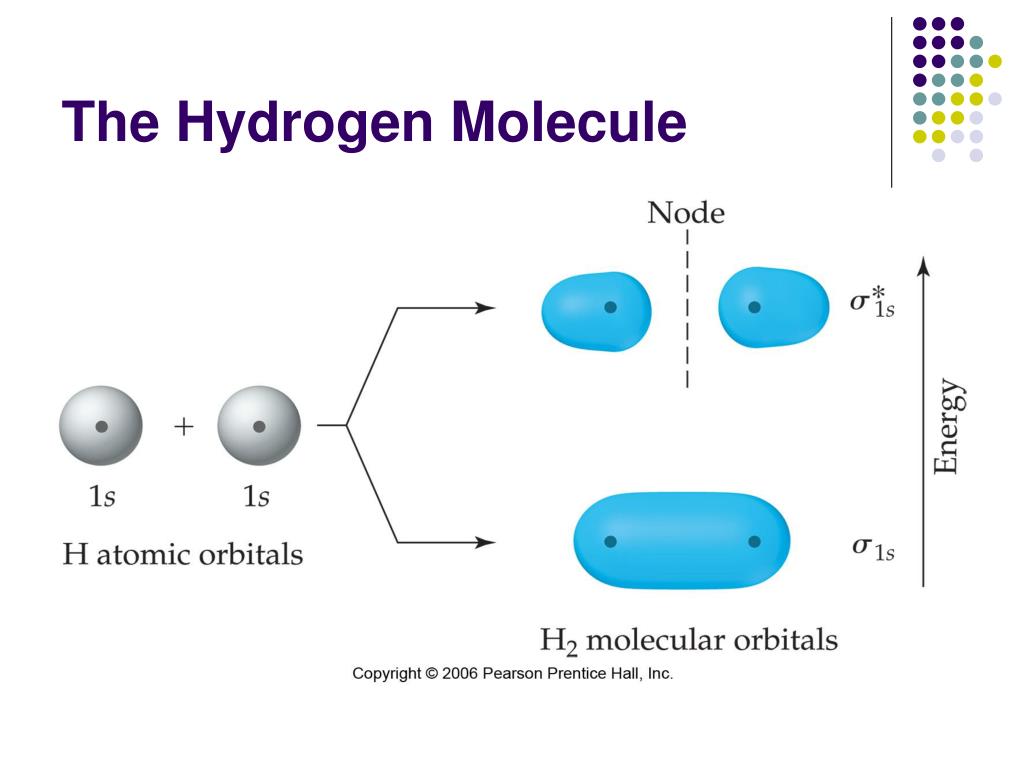
ISRO CS Syllabus for Scientist/Engineer Exam Combining the 1s orbitals of each hydrogen atom using LCAO, two molecular orbitals are generated 1s (pronounced sigma one s) and 1s (pronounced sigma.ISRO CS Original Papers and Official Keys.All orbitals from Helium and up are NOT Hydrogen. GATE CS Original Papers and Official Keys The atomic orbitals of Hydrogen are one of the few atoms that can be easily calculated in closed form.The letters come from old notation developed by spectroscopists, who noticed trends in atomic spectra well before quantum mechanics was developed to explain these trends. Notice that the energy level spacing decreases as n increases, that the number of orbitals (i.e. Full Stack Development with React & Node JS(Live) The orbital quantum number is usually specified indirectly in shorthand for the hydrogenic orbitals: 0 s, 1 p, 2 d, 3 f, 4 g. Lets look at the energy levels of the hydrogen atom.Atomic hydrogen constitutes about 75 of the baryonic mass of the universe. The electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the Coulomb force. OS DBMS CN for SDE Interview Preparation A hydrogen atom is an atom of the chemical element hydrogen.Full Stack Development with React & Node JS (Live).Data Structure & Algorithm Classes (Live).He found that the ratio of energy in electrons and the frequency of their orbits around the nucleus was equal to Planck's constant (the proportion of light's energy to its wave frequency, or approximately 6.626 x 10 - 23 ). Who discovered energy orbitals?īohr turned to Planck's quantum theory to explain the stability of most atoms. Which scientist found the orbitals?īohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element. He described it as the redistribution of the energy of orbitals of individual atoms to give new orbitals of equivalent energy and named the process as hybridisation. Scientist Pauling introduced the revolutionary concept of hybridization in the year 1931. Now, we have got the complete detailed explanation and answer for everyone, who is interested! FIGURE 22.1 Electron energy bands of liquid water made of atomic orbitals of hydrogen and oxygen 5 CB conduction band, VB valence band, sc > 1.2 eV, gv 9.3 eV, gg > 8 eV. This is a question our experts keep getting from time to time. The atomic orbitals of hydrogen are labeled by quantum numbers.Three integers are required for a complete specification. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
